|
12/20/2023 0 Comments Cathode anode battery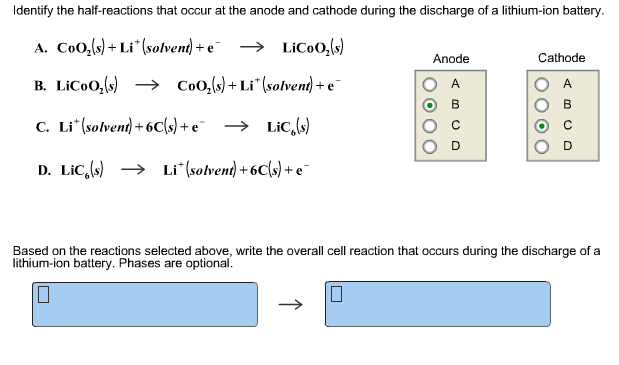
Simply put, the chemical reaction at the anode releases electrons and the reaction at the cathode absorbs them. Simultaneously, the cathode undergoes a reduction reaction wherein the material the cathode is made of, ions, and free electrons combine to form compounds. 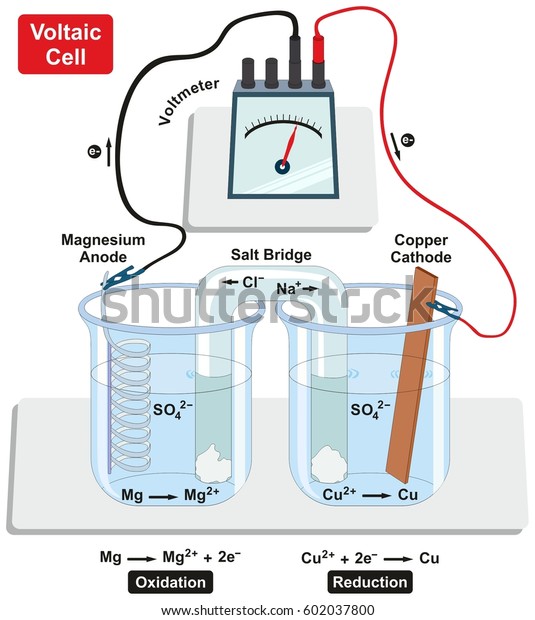
The anode undergoes what is known as an oxidation reaction: during discharge two or more ions from the electrolyte combine with the anode to form a compound and release one or more electrons. The energy is stored in the particular compounds that make up the anode, cathode and the electrolyte-for example, zinc, copper, and SO 4, respectively.Īssuming the battery has acquired its charged condition either by recharging or manufacturing, the aggregate effect of the chemical reactions taking place between the anode and the cathode discharges electricity. One electrode-the anode-permits electrons to flow out of it. The space between the electrodes is filled with an electrolyte-an ionic liquid that conducts electricity. Because the cell is where the actual action of storage and discharge takes place, this answer will concentrate on what happens at that level.Īll electrochemical cells consist of two electrodes separated by some distance. A battery is comprised of at least one but possibly many such cells appropriately connected. The former is the fundamental unit of electrochemical storage and discharge. In terms of storing energy or discharging electricity, they are similar, it is simply a question of whether or not the chemical processes involved permit multiple charging and discharging.īefore answering this question it is also necessary to distinguish between a galvanic cell and a battery, as I have defined it. There are two fundamental types of chemical storage batteries: the rechargeable, or secondary cell, and the non-rechargeable, or primary cell. Even within this restrictive definition, there are many possible chemical combinations that can store electrical energy-a list too long to go into in this short explanation. There are no batteries that actually store electrical energy all batteries store energy in some other form. These are the most common batteries, the ones with the familiar cylindrical shape. 
A battery for the purposes of this explanation will be a device that can store energy in a chemical form and convert that stored chemical energy into electrical energy when needed. There are a variety of chemical and mechanical devices that are called batteries, although they operate on different physical principles. First, the definition of a battery must be established. This question, which appears simple and direct, is actually filled with subtlety and complication. Kenneth Buckle, a visiting scientist at the Center for Integrated Manufacturing Studies at the Rochester Institute of Technology, provides this explanation.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
